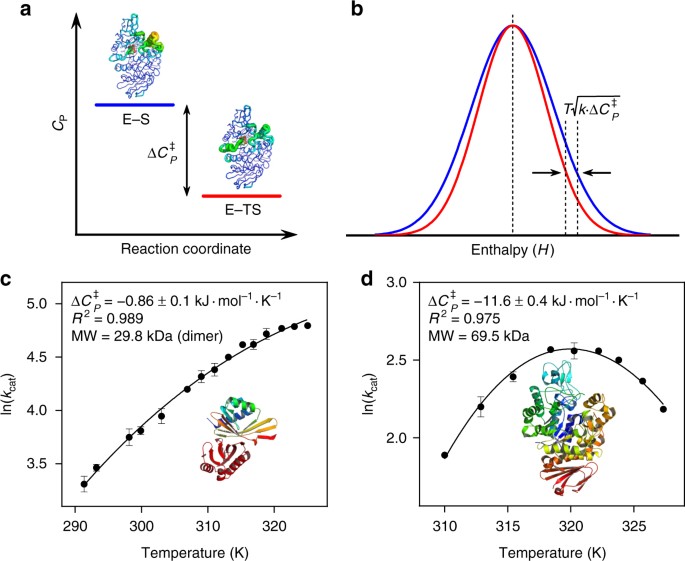
a) Activation energy ( Ea , kJ mol À 1 ), (b) Optimum temperature ( T... | Download Scientific Diagram

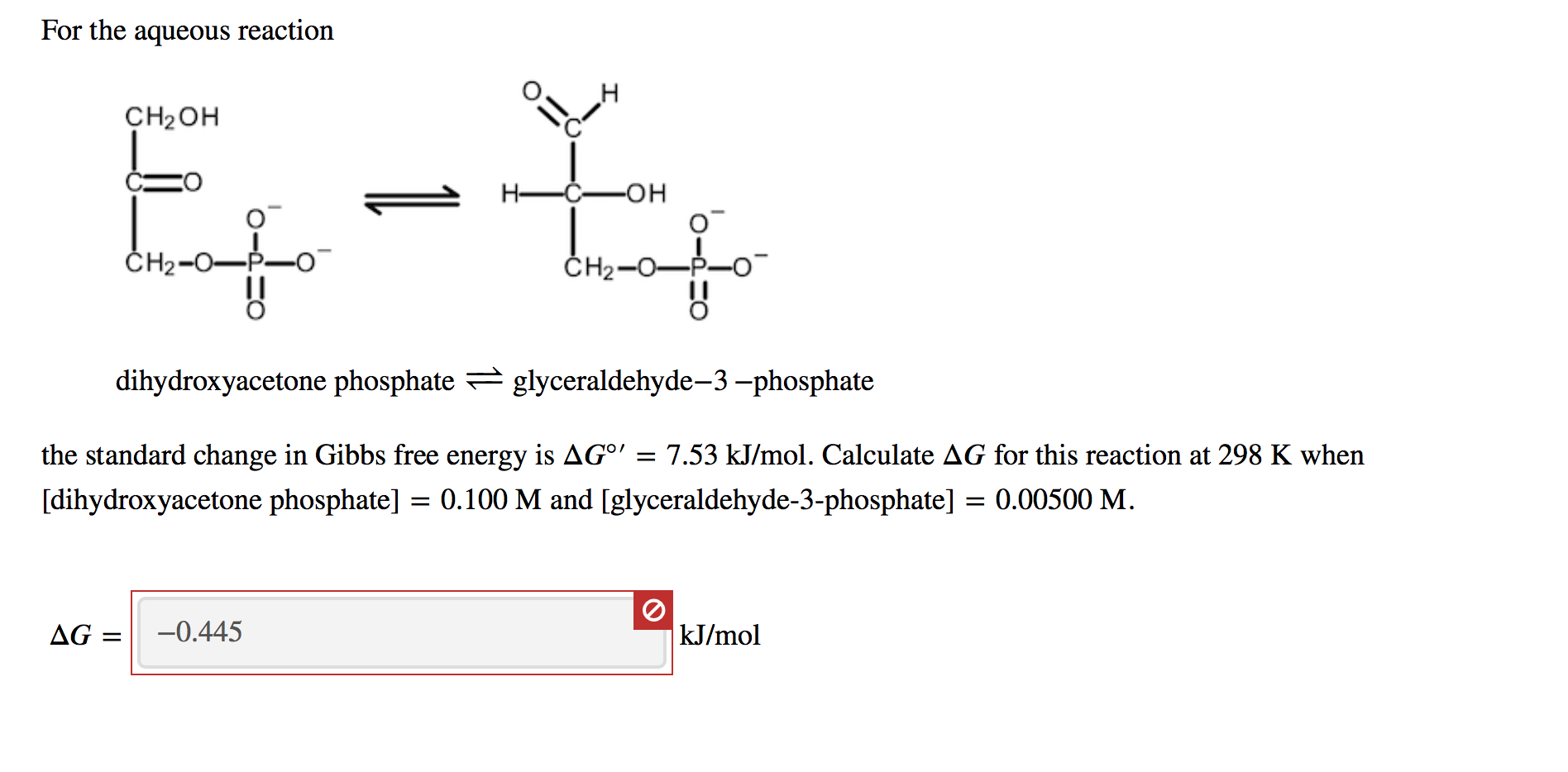
SOLVED: Consider a general reaction: enzyme A(aq) + B(aq) The ΔGo of the reaction is -9.990 kJ mol^-1. Calculate the equilibrium constant for the reaction at 25 °C. Keq = 54.36 What
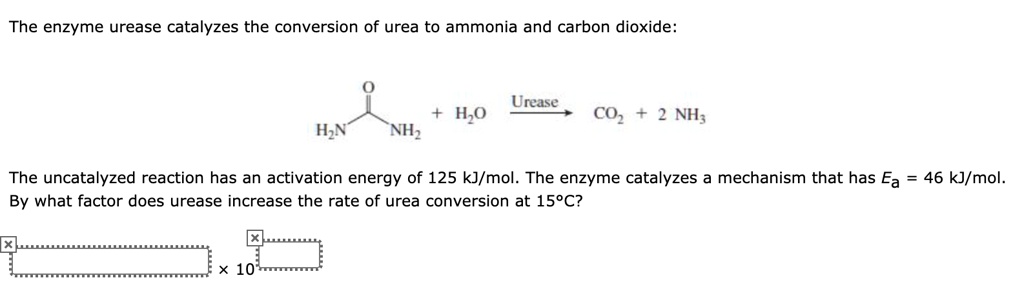
SOLVED: The enzyme urease catalyzes the conversion of urea to ammonia and carbon dioxide: Urease H,o COz 2 NH; HN NHz The uncatalyzed reaction has an activation energy of 125 kJ/mol. The

![Values of activation energy, E a [kJ mol -1 ] and Q 10 value for... | Download Scientific Diagram Values of activation energy, E a [kJ mol -1 ] and Q 10 value for... | Download Scientific Diagram](https://www.researchgate.net/publication/334326868/figure/tbl3/AS:1049389002936321@1627205243258/Values-of-activation-energy-E-a-kJ-mol-1-and-Q-10-value-for-several-reactions.png)