Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube
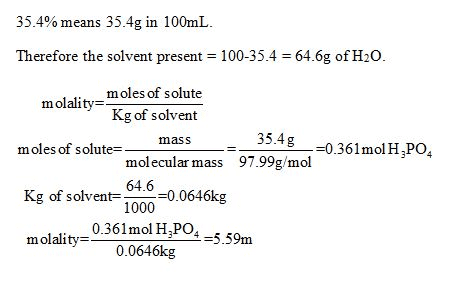
OneClass: Calculate molality of a 35.4% (by mass) aqueous solution of phosphoric acid (H3PO4). The mo...

Calculate molality of solution if the density of solvent is 1.2 × 10^3 kg/m^3 and molarity of solution is 0.5 molar.