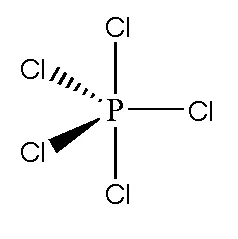
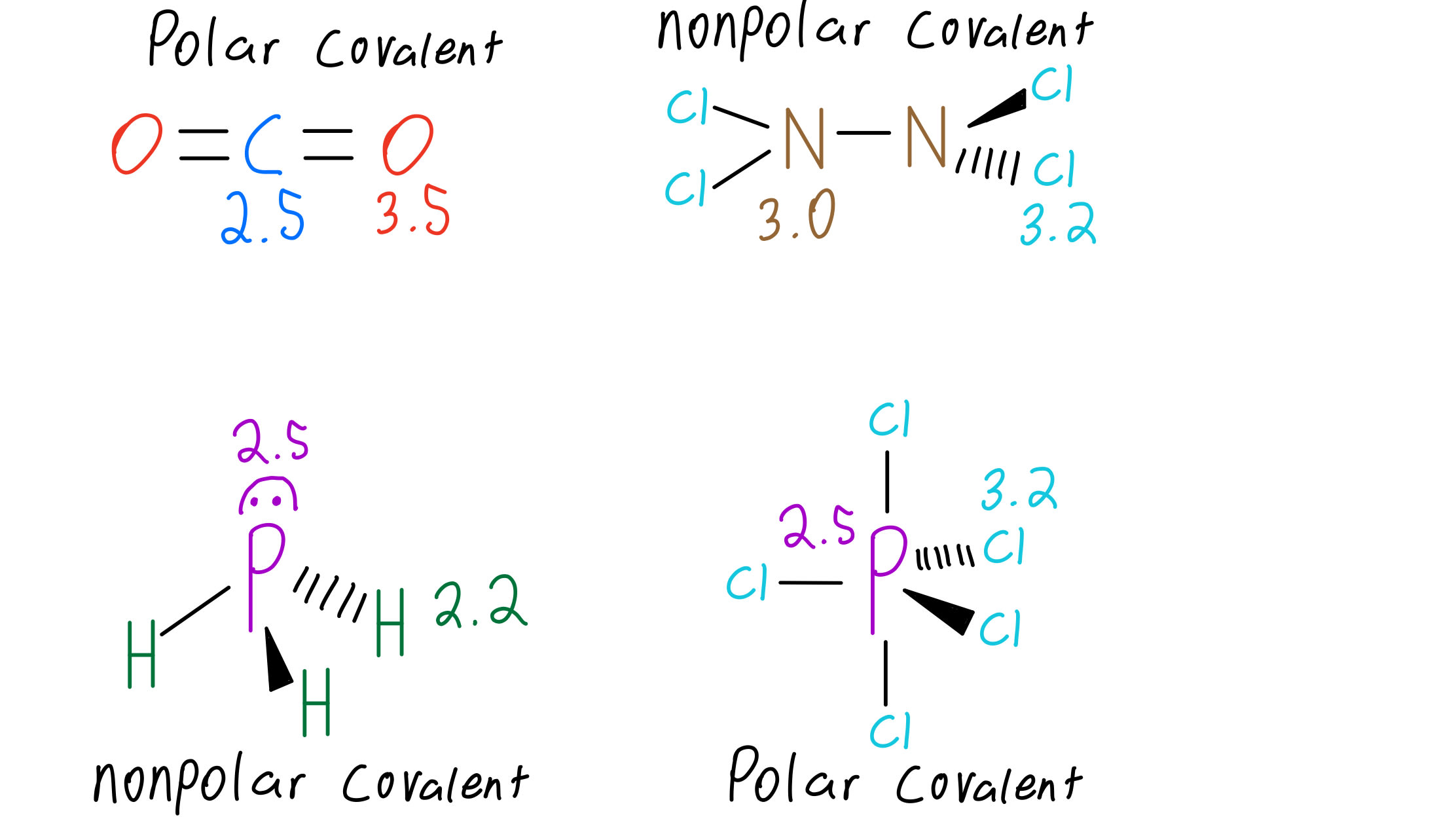By drawing the Lewis structures of the following molecules, determine which of them is polar. a. Cl2 b. Bh3 c. NH3 d. PCl5 | Homework.Study.com

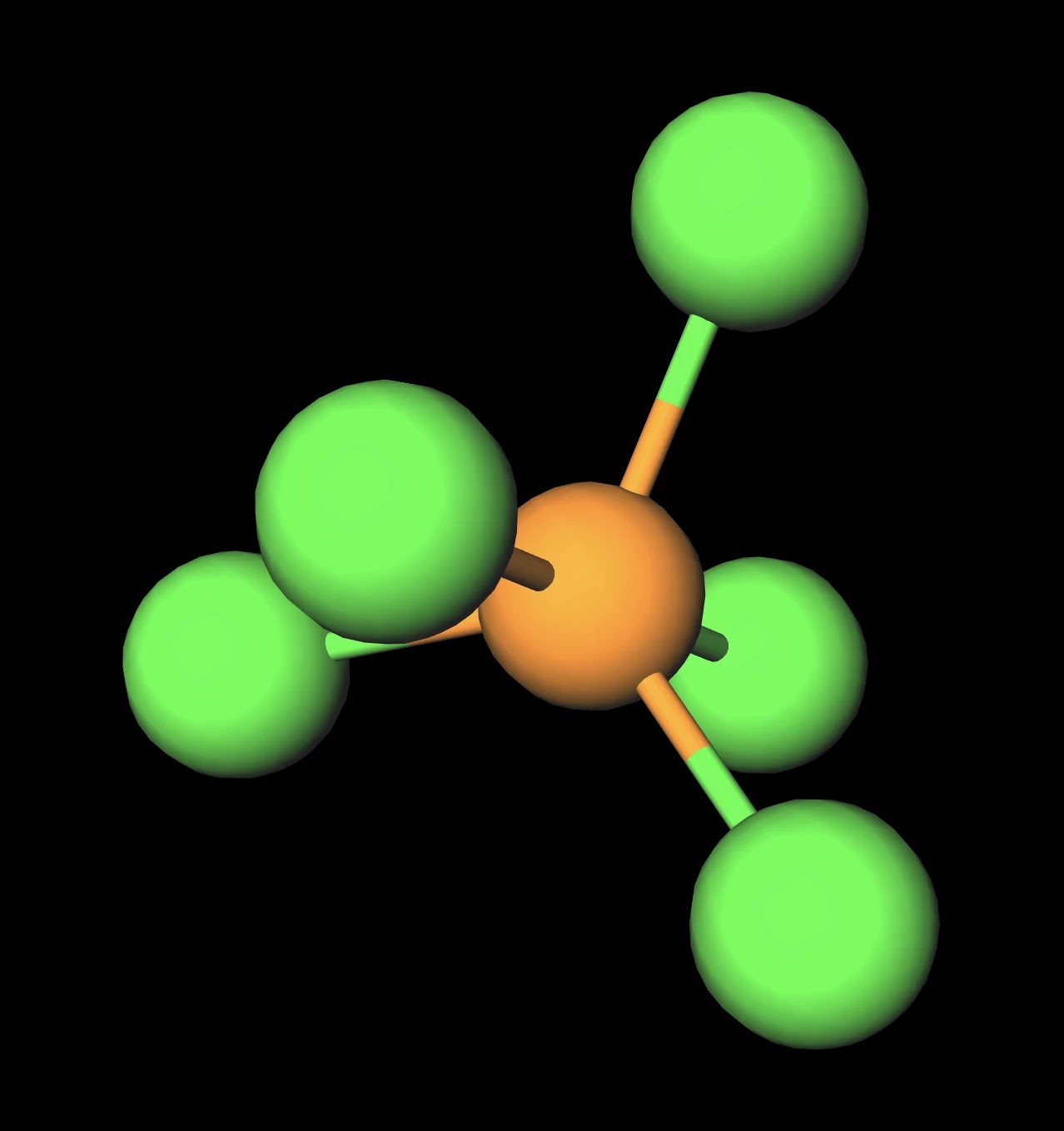
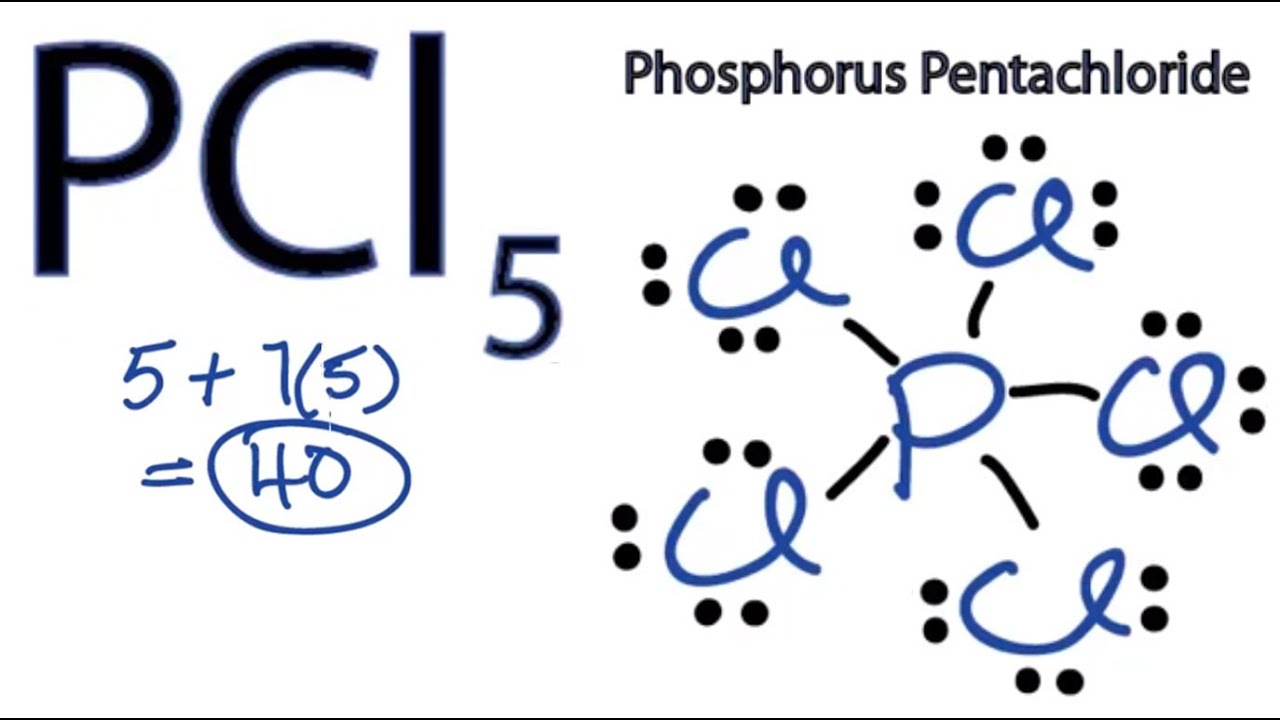
Draw the Lewis structure for PCl5 and answer the following questions. a. Does the central atom violate the octet rule? b. How many lone pairs of electrons are in the molecule? c.

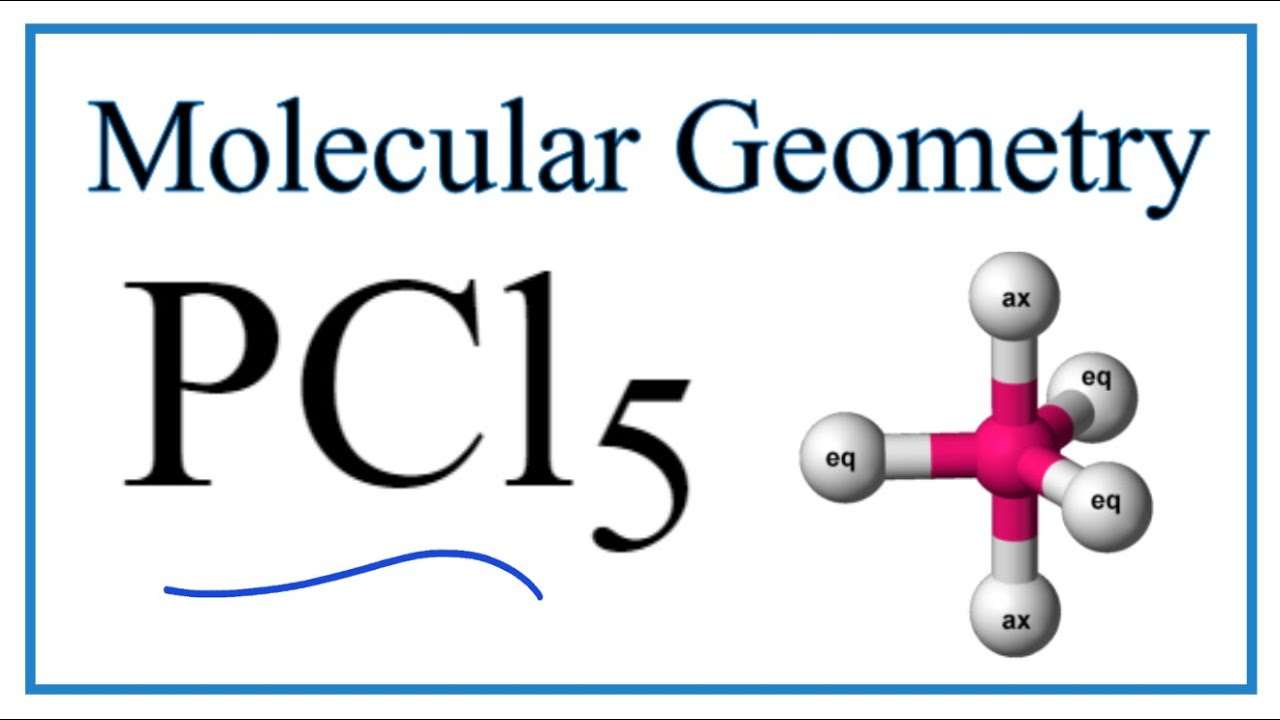
PCL5 Lewis Structure (Phosphorous Pentachloride) | PCL5 Lewis Structure (Phosphorous Pentachloride) Hi Everyone! Welcome back to our channel. In today's video, we help you determine the Lewis Structure... | By Geometry Of
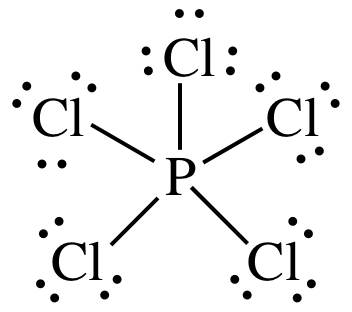
Section 11-1: Molecular Geometry: Using VSEPR Theory to Determine Three-Dimensional Shapes and Bond Angles